- You are here: >>
- Home >>
- Our Services >>
- Reverse Osmosis System Plants >>
REVERSE OSMOSIS SYSTEM
What is Reverse Osmosis System?
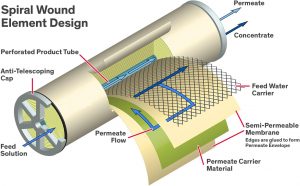
Reverse Osmosis System is a water purification technology that is used to remove a large majority of contaminants like fluoride ions, Nitrate molecules from water by pushing the water under pressure through a semipermeable membrane. A reverse osmosis system is built around its individual membranes. Each membrane is a spiral wound sheet of semi-permeable material. Membranes are available in 2-inch, 4-inch, and 8-inch diameter with the 4- and 8-inch diameter sizes most commonly used in industry. The industry has accepted a 40-inch length as a standard size so that membranes from different manufacturers are interchangeable in equipment systems. One of the primary measurements of a membrane is its square footage. Membranes are available in the range of 350-450 square feet of surface area. Semi-permeable membranes were first constructed using cellulose acetate (CA) but later the industry switched primarily to the use of a thin film composite (TFC) being placed on top of a stronger substrate. TFC membranes are primarily used today.
What is the role of RO Membrane?
When the water molecules pass through the membrane under very high pressure, they pass through it, and salts or contaminants hold back. They get discharged through the system by the reject stream. Rejected water may be drained out or sent to the feed water supply, depending on the design.
RO systems use Cross Filtration and not the standard filtration where the contaminants get accumulated in the filter media. In this case, the solution passes through the filter in two different ways. Contaminated water goes in one direction and the filtered water in other. Cross flow filtration cleans the contaminant build up so that the membrane surface remains clean.
How Does Reverse Osmosis Work?
Diffusion is the movement of molecules from a region of higher concentration to a region of lower concentration. Osmosis is a special case of diffusion in which the molecules are water and the concentration gradient occurs across a semipermeable membrane. The semipermeable membrane allows the passage of water, but not ions (e.g., Na+, Ca2+, Cl-) or larger molecules (e.g., glucose, urea, bacteria). Diffusion and osmosis are thermodynamically favorable and will continue until equilibrium is reached. Osmosis can be slowed, stopped, or even reversed if sufficient pressure is applied to the membrane from the ‘concentrated’ side of the membrane.
Reverse Osmosis System
Reverse osmosis occurs when the water is moved across the membrane against the concentration gradient, from lower concentration to higher concentration. To illustrate, imagine a semipermeable membrane with fresh water on one side and a concentrated aqueous solution on the other side. If normal osmosis takes place, the fresh water will cross the membrane to dilute the concentrated solution. In reverse osmosis, pressure is exerted on the side with the concentrated solution to force the water molecules across the membrane to the fresh water side.
Reverse osmosis is the process of forcing a solvent from a region of high solute concentration through a membrane to a region of low solute concentration by applying a pressure in excess of the osmotic pressure. This is the reverse of the normal osmosis process, which is the natural movement of solvent from an area of low solute concentration, through a membrane, to an area of high solute concentration when no external pressure is applied. The membrane here is semipermeable, meaning it allows the passage of solvent but not of solute.
To illustrate, imagine a semi permeable membrane with fresh water on one side and a concentrated aqueous solution on the other side. If normal osmosis takes place, the fresh water will cross the membrane to dilute the concentrated solution. In reverse osmosis, pressure is exerted on the side with the concentrated solution to force the water molecules across the membrane to the fresh water side.
The membranes used for reverse osmosis systems have a dense polymer barrier layer in which separation takes place. Since Reverse Osmosis does not occur naturally, it must be created by applying pressure to the high solids water in order to force it through the membrane, with pressures from 8 – 14 bar for fresh and brackish water, and 40 – 70 bar for seawater, which has around 24 bar (350 psi) natural osmotic pressure which must be overcome.
Design Considerations for RO Plant
Fluid Systems RO Plants work on the Cross flow Filtration method, which takes the feed water and uses a percentage of it as a wash or reject stream, removing the solids during the filtration process. The product flow of an RO Plant is mainly a function of temperature and pressure. System recovery (product divided by feed) is limited by the characteristics of the feed water and can be controlled through the use of recycle stream. Product quality is based on a percentage of dissolved solids fed to the membrane. There should be an economic balance between product quality and system recovery. High recoveries increase concentration of dissolved solids in the system which degrades quality, but high recoveries make the system work more efficiently and decrease waste.
Reverse Osmosis Plants do not deliver to service all of the water that is fed to them. During operation, some of the incoming water is used to wash down the membrane, and only part becomes finished product water. Purified water is referred to as product and wastewater is referred to as concentrate, or reject. The percent of water delivered as product is called the recovery, and depends upon the membrane and on total RO Plant design considerations.
Factors Affecting Reverse Osmosis System and Performance:
- The performance of a system depends on factors such as membrane type, flow control, feed water quality, temperature and pressure.
- Also only part of the water entering the unit is usable, this is called the % recovery.
- For example, the amount of treated water produced can decrease by about 1-2% for every 1 degree Celsius below the optimum temperature.
- Systems must be well maintained to ensure good performance with any fouling requiring cleaning maximizing the output of water.
- Biocides may be needed and the choice of biocide would depend on the membrane type, alternatively, other filters may be required to remove chlorine from water to protect the life of the membranes.
- To this end, a good treatment regime is needed and knowledge of the specific foulants so the optimum cleaning and maintenance chemicals can be chosen.
What contaminants will Reverse Osmosis remove from water?
The Normal water consists of Fluorides, Chlorine & chloramine, Lead, Detergents & more Nitrates & Sulfates Reverse Osmosis is capable of removing up to 99%+ of the dissolved salts (ions), particles, colloids, organics, bacteria and pyrogens from the feed water (although an RO system should not be relied upon to remove 100% of bacteria and viruses).
An RO membrane will reject every contaminants based on their size and charge. Any contaminant that has a molecular weight greater than 200 is likely rejected by a properly functioning RO system.For example, a monovalent sodium ion and divalent calcium will not be rejected by the RO. That is the Why an RO system cannot separate gases like Carbon Dioxide (Co2).Reverse Osmosis is very effective for treating brackish, surface and ground water for both large and small flows applications. Some of industries that use RO water include pharmaceutical, boiler feed water, food and beverage, metal finishing and semiconductor manufacturing to name a few.
Effectiveness of RO System
We being the reverse osmosis plant manufacturers and suppliers pride ourselves to be considered as one of the best in town.
- It can remove up to 99% organics, particles, ions, pyrogens, and bacteria. However, it doesn’t claim 100% removal of bacteria and viruses.
- Contaminants are rejected based on their molecular weight and size. Typically, particles of molecular weight more than 200 get rejected by a well-maintained RO system. To give an example
- It also identifies contaminants based on the ionic charge. Molecules with a high ionic charge will not pass through the membrane. Gases such as CO2 don’t have highly charged atoms, and their molecular weight is also low. Hence, they pass through the membrane. It is the reason; RO water has higher pH value.
Reverse Osmosis is a multipurpose, versatile method of purifying water. It can treat surface and ground water very effectively. It can treat brackish water also. RO is ideal for both large and small flow applications.
Industries such as boiler feed, pharmaceutical, semiconductor manufacturing, metal finishing food and beverage widely use RO water. They have requirement of huge RO treatment plants that Cleantech is capable of providing. Our experts work with the clients to understand their needs. We suggest the best solution.
A few parameters determine the efficiency and usability of RO systems. The quality of water, flow, and pressure are some important aspects. Also, hours of operations, the temperature of water, feed pressure, permeate pressure, and concentrate pressure are also important. Feed and permeate conductivity, feed and permeate flow, and salt rejection percentage are some other parameters.
Due to excellent performance and quality of the output water, good quality Reverse Osmosis Plants is in high demand. We have the complete range of choices to the customers spread across industries.
Austro One Stop Solution for All your Drinking Water Problems
Austro is one of south India’s trusted Industrial RO Plant Manufacturers. We have designed and commissioned more than 85 ETP plants in South India Region. We have a proven expertise of drinking water treatment. As Reverse osmosis system is the most preferred method for water purification. We offer solutions for Reverse osmosis plant with a help of systematic plant installation. We can also provide you an effective plant maintenance guidance. We aim on delivering best quality RO installation.
Advantages of Using Reverse Osmosis System
- Simple to operate.
- Energy efficient, especially when used instead of distillation to produce high purity water
- Does not require hazardous chemicals.
- The processing system is simple; the only complicating factor is finding or producing a clean supply of feedwater to minimize the need for frequent cleaning of the membrane.
- Systems may be assembled from prepackaged modules to produce a supply of product water ranging from a few liters per day to 750 000 l/day for brackish water, and to 400 000 l/day for seawater; the modular system allows for high mobility, making RO plants ideal for emergency water supply use.
- Installation costs are low.
- RO plants have a very high space/production capacity ratio, ranging from 25 000 to 60 000 l/day/m2.
- Low maintenance, nonmetallic materials are used in construction.
- Energy use to process brackish water ranges from 1 to 3 kWh per 1 0001 of product water.
- RO technologies can make use of use an almost unlimited and reliable water source, the sea.
- RO technologies can be used to remove organic and inorganic contaminants.
- Aside from the need to dispose of the brine, RO has a negligible environmental impact.
- The technology makes minimal use of chemicals.
Benefits of Industrial Reverse Osmosis System
- Reduces water and sewer use costs.
- Can be integrated with an ion exchange system to achieve up to 80% rinse water recycle.
- Modular design for ease of installation
Disadvantages of Using Reverse Osmosis Membrane:
- The membranes are sensitive to abuse.
- The feed water usually needs to be pretreated to remove particulates (in order to prolong membrane life).
- There may be interruptions of service during stormy weather (which may increase particulate resuspension and the amount of suspended solids in the feed water) for plants that use seawater.
- Operation of a RO plant requires a high quality standard for materials and equipment.
- There is often a need for foreign assistance to design, construct, and operate plants.
- An extensive spare parts inventory must be maintained, especially if the plants are of foreign manufacture.
- Brine must be carefully disposed of to avoid deleterious environmental impacts.
- There is a risk of bacterial contamination of the membranes; while bacteria are retained in the brine stream, bacterial growth on the membrane itself can introduce tastes and odors into the product water.
- RO technologies require a reliable energy source.
OUR APPLICATION
- Ceramic.
- Chemicals
- Dairy
- Food & Beverages
- Hospitals
- Pharmaceuticals
- Hotel
- Pulp & Paper
- Textile




No Comments
Sorry, the comment form is closed at this time.